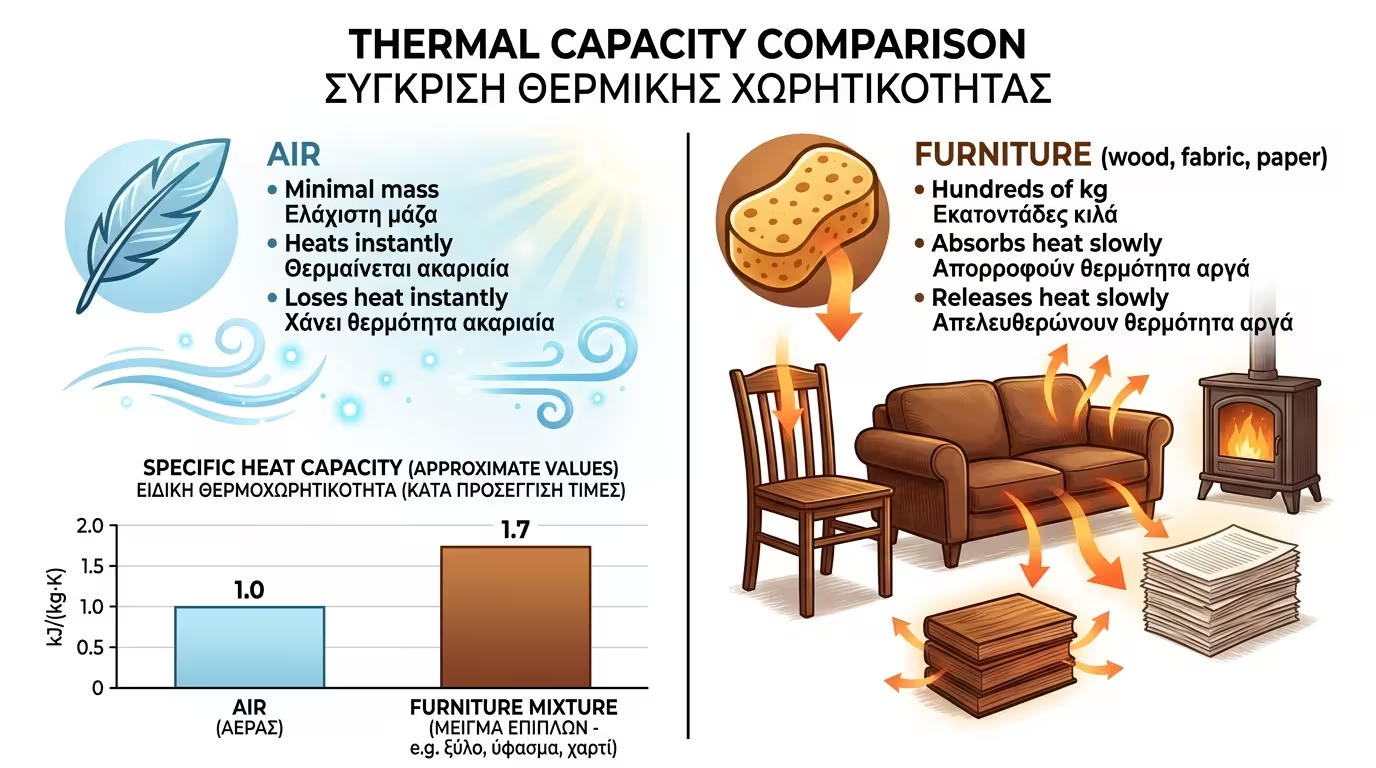
💨 Air
An extremely light gas. It has minimal mass. It heats instantly but loses its heat just as instantly. It cannot "store" anything.
Have you ever entered a brand-new, completely empty (unoccupied) house in winter? Even though it may have perfect insulation and new windows, as soon as you turn on the heating something strange happens: the air warms quickly, but the moment you turn off the radiator or open the door for seconds, the house "freezes" instantly.
In contrast, in the house you already live in, the temperature seems to have a sweet "stability". What is to blame? The empty house is missing the biggest, free heat battery you own: your furniture.
To understand what happens, we must recall the concept of Thermal Capacity (Mass) we saw in a previous article. It is a material's ability to store heat.
An extremely light gas. It has minimal mass. It heats instantly but loses its heat just as instantly. It cannot "store" anything.
A huge corner sofa, a massive wooden bookcase full of books (paper), the wooden table, heavy rugs and beds, weigh hundreds of kilograms in total. Wood, fabric and paper have enormous specific heat capacity.
When you turn on the radiator, you are not just heating air. Thermal energy is gradually transferred to the furniture. Your sofa and books "soak up" the heat like sponges.
This energy storage is what makes the difference in comfort and savings:
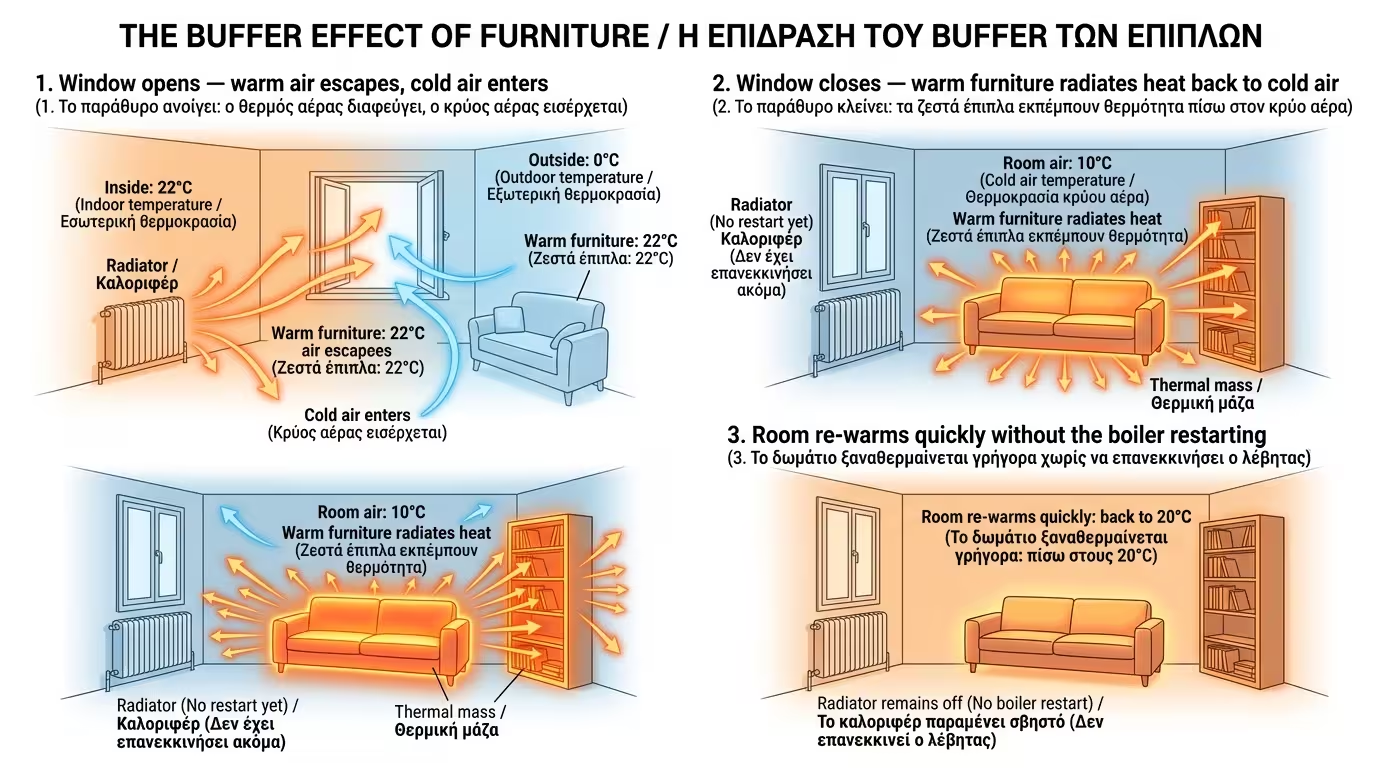
In winter, if you open the window for 5 minutes to air out, all the warm air leaves. If the house were empty, the boiler would have to start from scratch. In the furnished house, as soon as you close the window, the warm furniture (the battery) immediately begins radiating its heat back into the new, cold air. The room warms up quickly without the boiler kicking in!
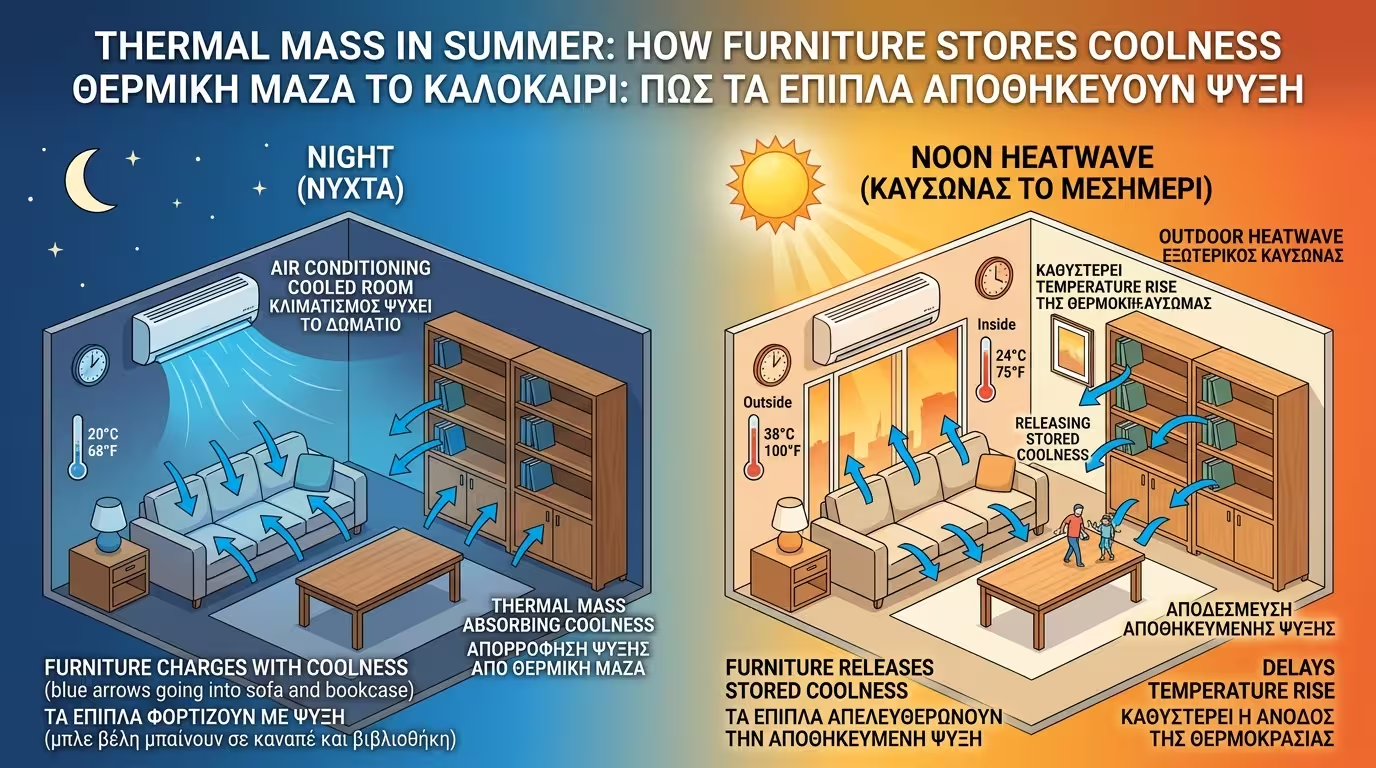
The same applies in summer. If you cooled the house with the air conditioner all night, the furniture "charges" with coolness (releases its heat). At noon when the heatwave hits, the heavy, cool sofa and bookcase will delay the temperature rise in the room.
We have our digital house of 100 m², containing 300 cubic metres of air. It is winter.
The house has not a single chair. We turn on a fan heater. Because we are only heating air (minimal mass), the thermometer hits 22°C in just 15 minutes. Perfect! We turn off the heater. In 20 minutes, the heat has escaped to the walls and the thermometer drops to 17°C. The system must run continuously.
The house has 1,500 kg of furniture, books and rugs. We turn on the heating. The thermometer takes a long time to reach 22°C (maybe 2 hours), because a large part of the energy goes to "warming" the sofa and the dining table. But once it hits 22°C and we turn off the heating, the house stays warm for the next 4 hours! The air starts to cool, but the furniture returns the heat it had absorbed.
💡 Final Conclusion: Never underestimate the mass of objects inside your home. A large bookcase on a north wall literally acts as an extra layer of insulation and thermal capacity. When you do energy calculations (or when you enter a new, empty house and are disappointed by the cold), remember that once you place your belongings, the space's thermal behaviour will improve dramatically!
Return to category.
Go to categoryReturn to the central guide.
Go to guide