🛡️ Passive Protection (Barrier Effects)
The primer creates a physical obstacle preventing water and oxygen contact with the metal. Materials such as Micaceous Iron Oxide (MIO) enhance this phenomenon by creating a "laminar" flake structure.
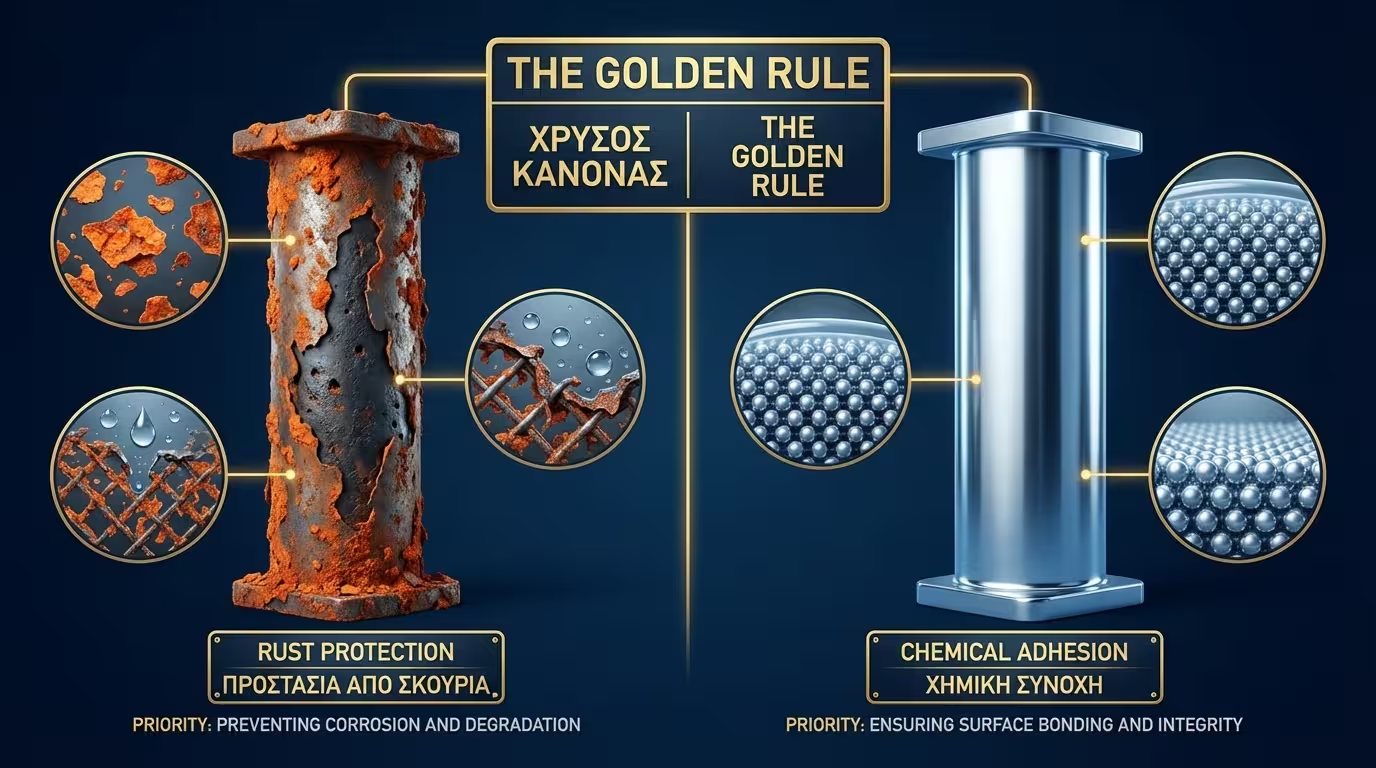
Primer selection is not merely a matter of adhesion, but the creation of a chemical or electrochemical shield that determines the lifespan of the entire metal structure.
In coating engineering, the primer is selected based on the metal substrate and the exposure environment. The goal is twofold: ensuring wettability for perfect adhesion and inhibiting the anode of the galvanic cell.
The primer creates a physical obstacle preventing water and oxygen contact with the metal. Materials such as Micaceous Iron Oxide (MIO) enhance this phenomenon by creating a "laminar" flake structure.
These primers contain pigments (e.g., Lead-free Minium, Zinc Phosphate) that react chemically with the surface, creating a passivation layer that "deactivates" corrosion.
Each chemical family of primers serves a specific industrial or construction purpose:
The economical and "forgiving" solution for ferrous metals. Due to slow drying, they possess excellent surface penetration on metals with residual oxidation. Ideal for railings and rural applications.
Specialized systems based on phosphoric acid. They perform chemical etching on the non-ferrous metal surface, creating a phosphate salt layer that guarantees adhesion.
The ultimate industrial "barrier". They offer superior chemical resistance and zero moisture permeability. Essential for tanks, ships, and structures in C4/C5 environments.
Even the best primer will fail if the surface has "low energy" due to contaminants. Alkaline cleaning or solvent degreasing is the step that determines if adhesion will be molecular or merely superficial.
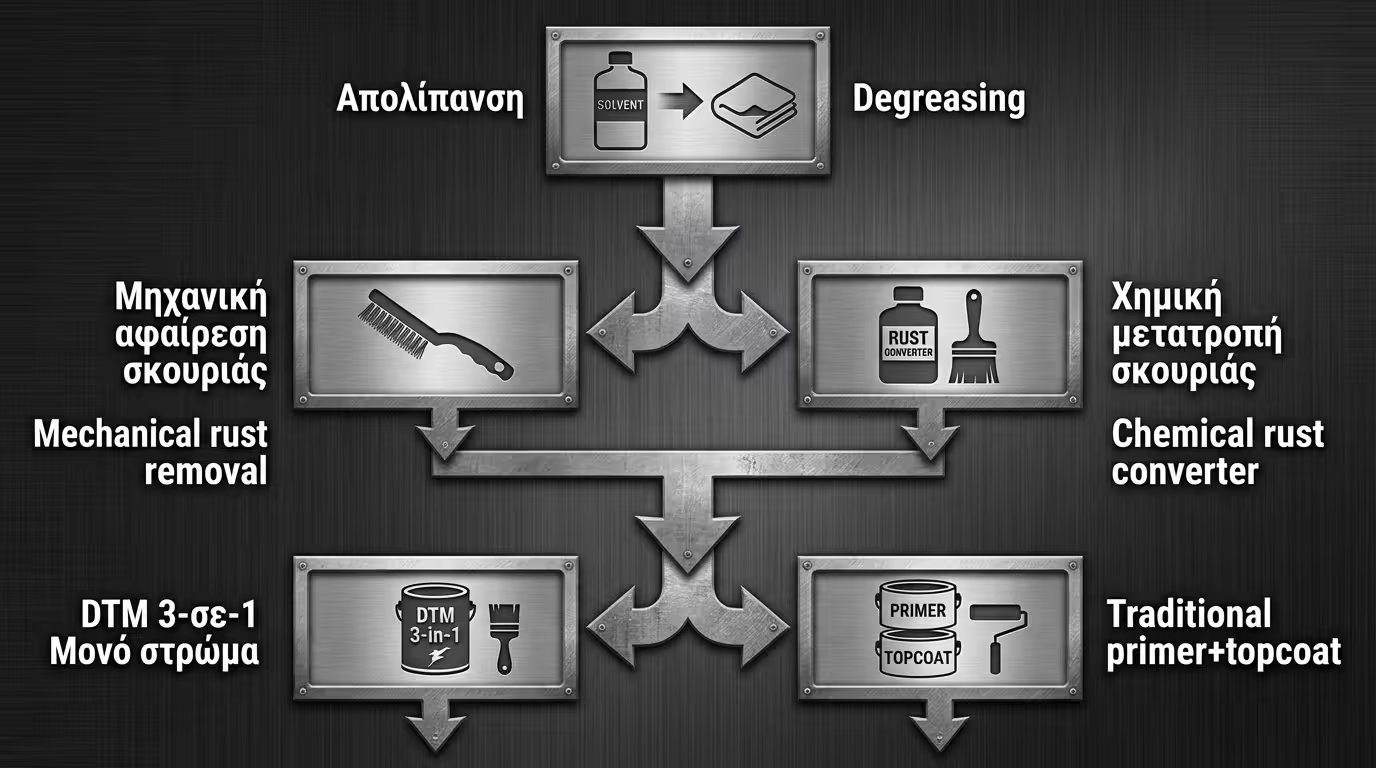
On new steel, the black oxide layer is brittle. If not removed mechanically, the primer will stick to the scale rather than the metal, leading to catastrophic delamination.
Converters are a compromise. In serious applications, removing the oxide to "white metal" and applying a Zinc-Rich Prime for electrochemical action is preferred.
In metal projects, failure always begins at the edges. Stripe Coating is the application of an extra primer coat via brush specifically to corners and welds before the main surface is sprayed.
A professional doesn't "look" at the paint; they measure it. Using magnetic gauges ensures the primer meets the minimum thickness (e.g., 75μm) required for anti-corrosive functionality.
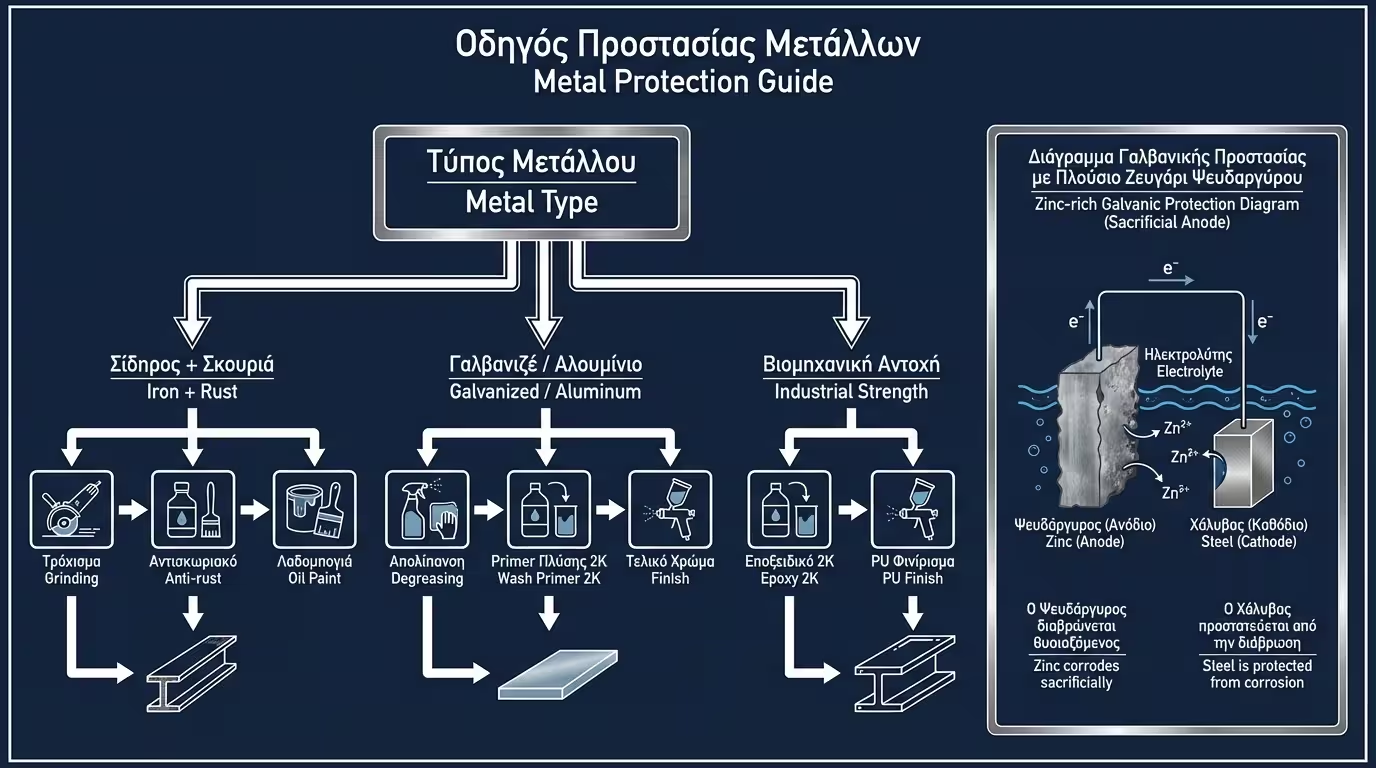
In high-salinity environments, zinc-rich primers act as a sacrificial anode. Even if the surface is scratched, the zinc "dies" first, protecting the steel substrate.
💡 Engineer's Verdict: For maximum service life, the "2K Epoxy Primer + Polyurethane Topcoat" system is the Golden Standard, as it combines absolute sealing with UV protection.
Return to category.
Go to categoryReturn to the central guide.
Go to guide