🧱 Cast Iron
Contains >2% carbon. It is brittle and oxidizes rapidly and deeply. Its coarse surface "absorbs" paint due to high porosity.
Why paint reacts differently on cast iron compared to carbon steel.
While we often use the terms interchangeably, from a metallurgical standpoint, cast iron and steel are distinct materials. Iron is more porous and coarse, whereas steel is smooth and elastic. These differences dictate paint adhesion and the overall lifespan of the coating.
Contains >2% carbon. It is brittle and oxidizes rapidly and deeply. Its coarse surface "absorbs" paint due to high porosity.
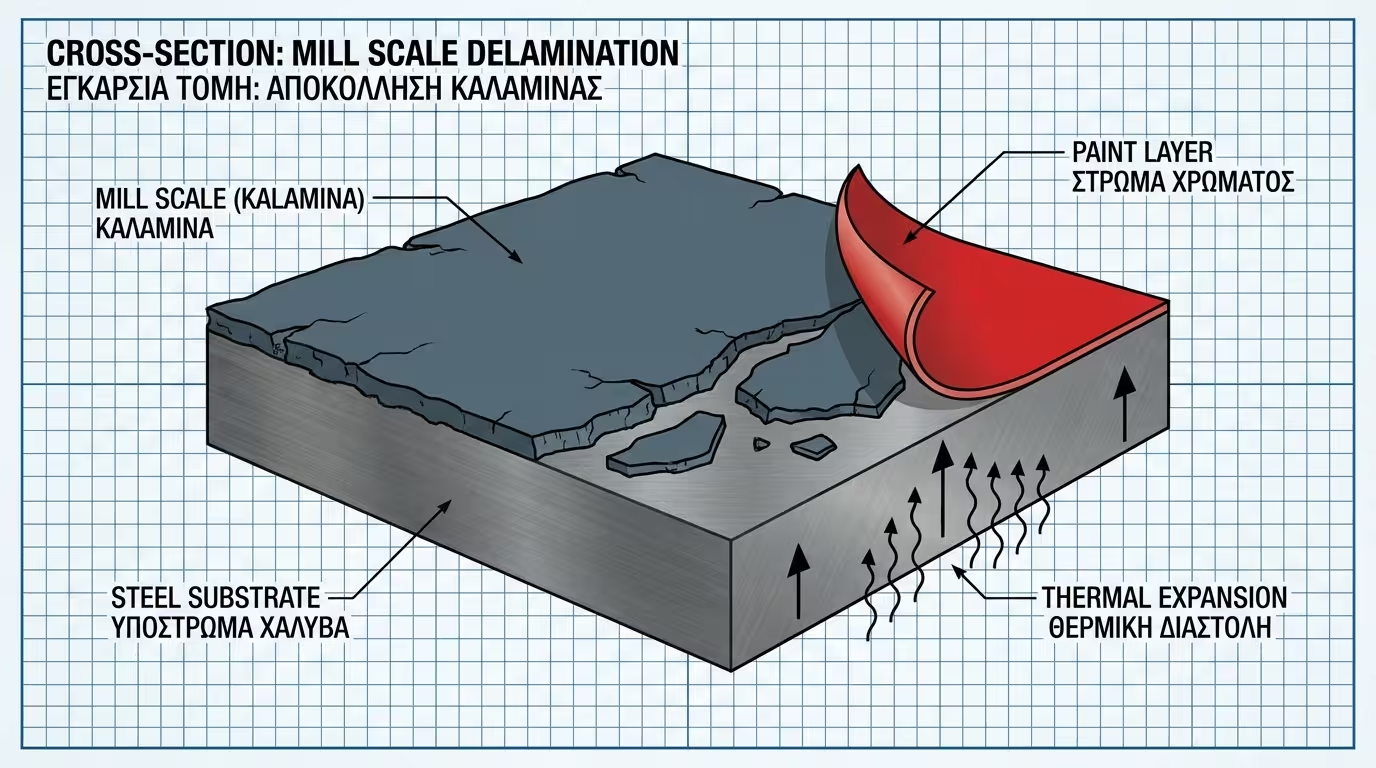
Iron-carbon alloy (<2.1%). It's the ultimate construction material, but it hides the trap of Mill Scale.
New carbon steel is covered in Mill Scale, a hard, blue-black oxide layer. It may look protective, but it has zero adhesion to the metal core.
If you paint over it, as soon as the metal expands or contracts, the mill scale will pop off, taking the entire paint system with it. Removing it via sandblasting or vigorous grinding is mandatory.
For maximum protection, especially in marine environments, Zinc-rich primers are used. These primers function as a sacrificial anode.
The zinc within the primer oxidizes instead of the steel. Even if the paint is scratched, rust cannot spread underneath the film.
Two-component epoxy systems offer maximum chemical and mechanical resistance, "locking" the metal structure.
📐 Engineering Tip: On carbon steel, sandblasting to Sa 2.5 grade is the only guarantee for permanent adhesion of high-performance epoxy systems.
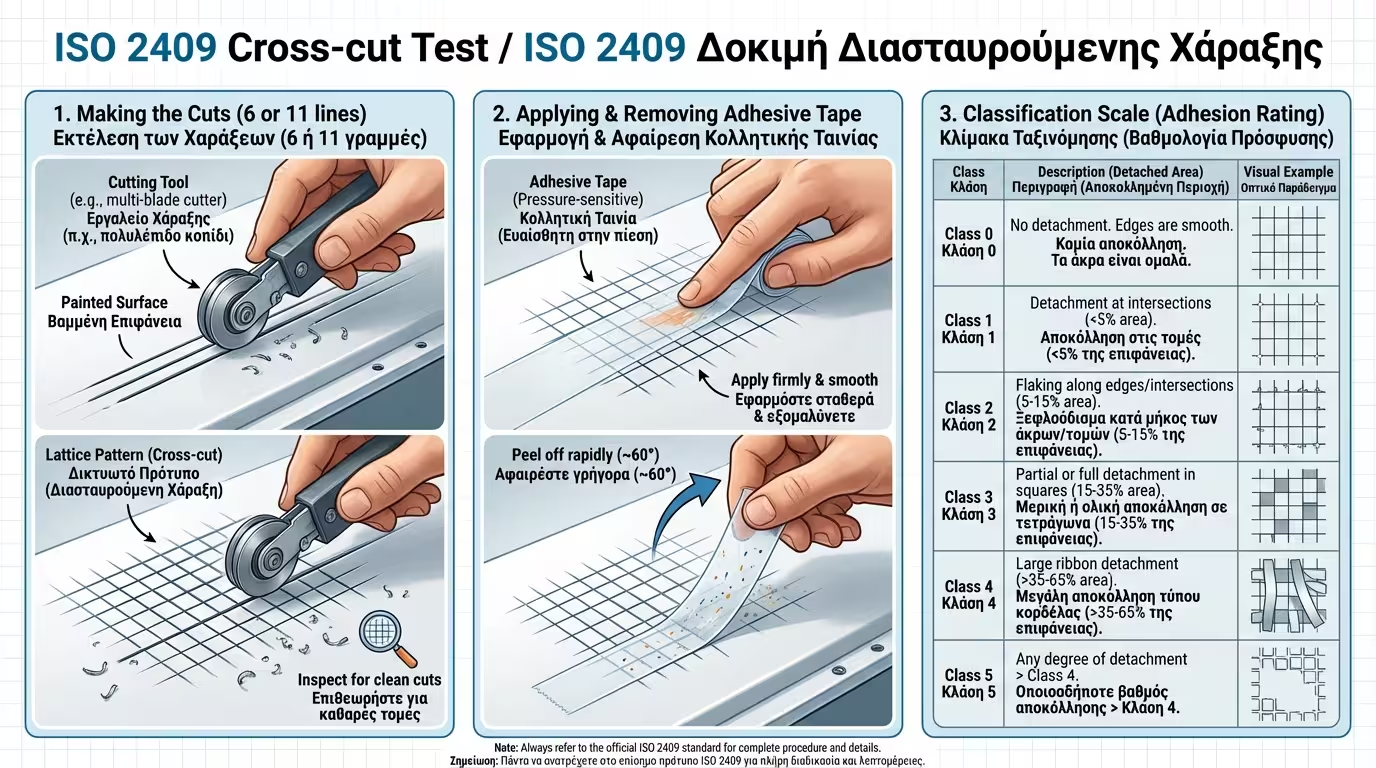
Before repainting, we use the Cross-cut test (ISO 2409). We carve a grid into the old paint and use specialized tape to see if it peels off.
If peeling exceeds 15% (Class 3-5), the old paint must be completely removed. If it's below 5%, it is safe to paint over.
Return to category.
Go to categoryReturn to the central guide.
Go to guideReturn to the main metal painting guide.
View morePlanning to paint a large metal structure?
Contact us