🛠️ St 2 / St 3 Cleaning (Hand & Power Tools)
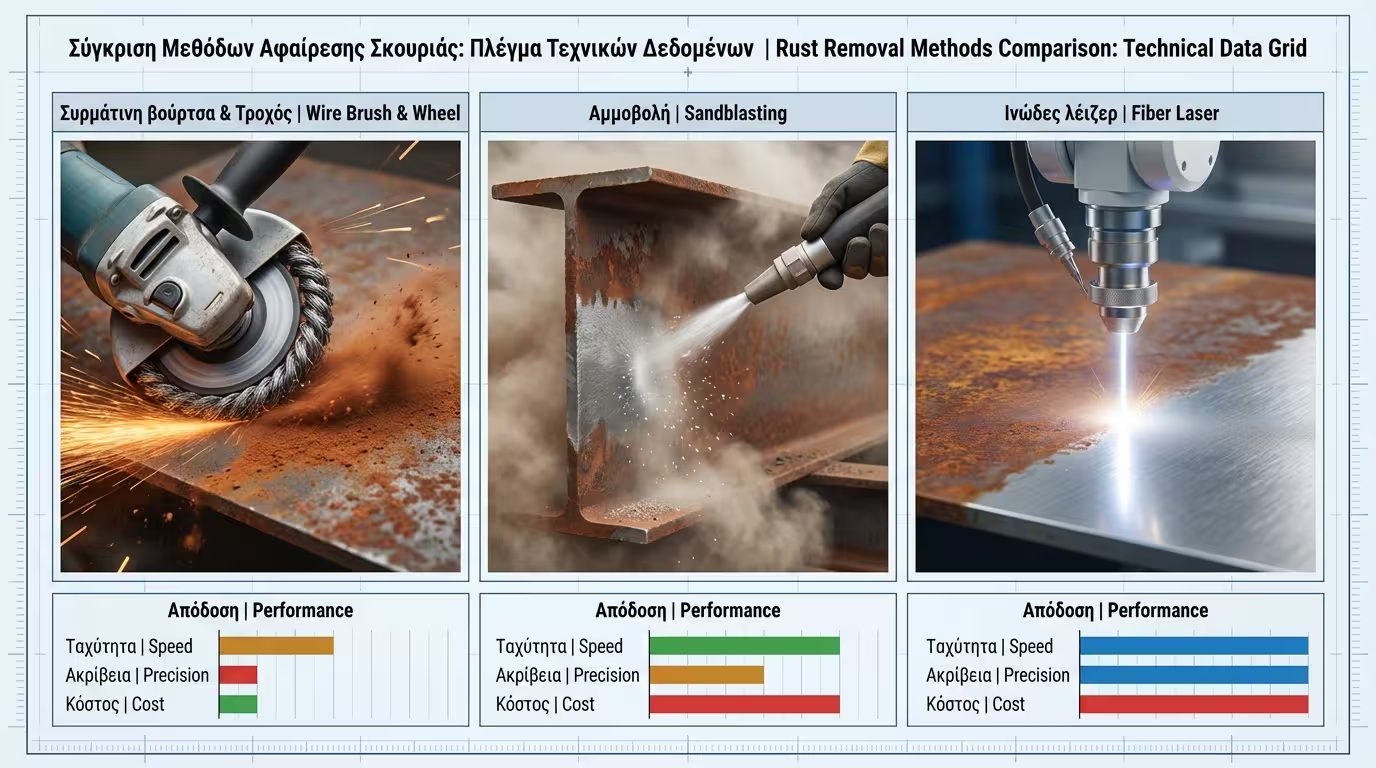
Manual or mechanical sanding. St 3 (very thorough) requires the surface to attain a metallic sheen. It is the "realistic" solution for home railings but leaves micro-traces of rust in the pores.
Rust is not just surface discoloration; it is an active electrochemical circuit. Permanently stopping it requires adherence to international industrial standards (ISO 12944) and the correct choice between mechanical removal and chemical conversion.
The effectiveness of any coating depends on the degree of cleanliness and the anchor profile (surface roughness) created on the metal.
Manual or mechanical sanding. St 3 (very thorough) requires the surface to attain a metallic sheen. It is the "realistic" solution for home railings but leaves micro-traces of rust in the pores.
The industrial "gold standard". Removes 95% of contaminants, creating a uniform profile of 50-75μm that mechanically "locks" the primer to the metal.
Cutting-edge technology. The laser uses pulse ablation to vaporise rust without affecting the steel's crystal lattice. Ideal for high-precision restoration without abrasive waste.
In areas with difficult access (e.g., metal joints), chemistry is the only solution. A converter does not remove rust but passivates it, turning it into a stable substrate.
Tannic acid reacts with iron oxide to create a black, insoluble polymer complex. This layer acts as a permanent moisture barrier.
Using a converter on stable rust increases epoxy system adhesion by up to 6 times, as the resin "anchors" into the micro-cracks of the transformed layer.
To stop rust permanently, we must intervene in the electrochemistry of the surface.
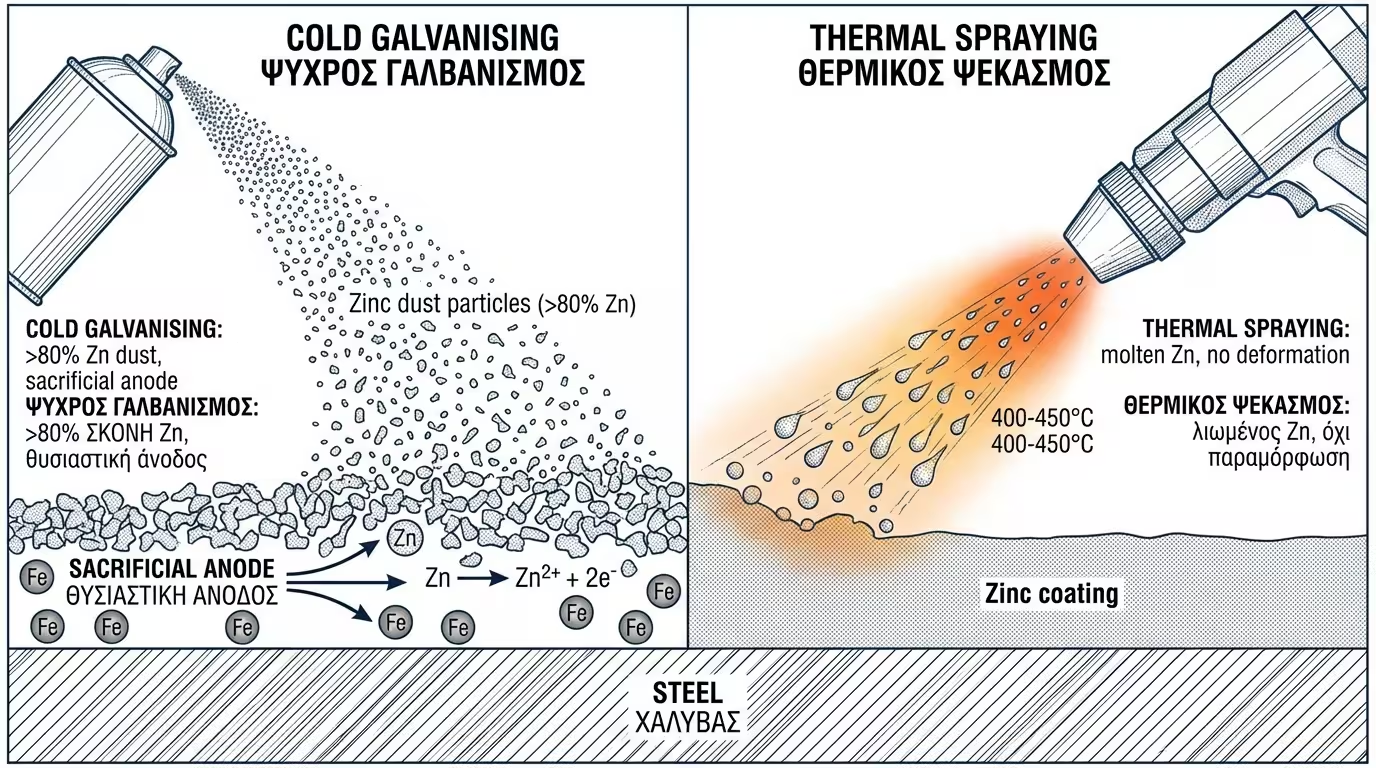
Contain >80% zinc dust. They act as a sacrificial anode: the zinc oxidises instead of the steel. Even if the coating is scratched, rust won't "creep" under the film.
Projecting molten zinc. Offers the protection of hot-dip galvanising without the risk of thermal distortion (warping) of the structure.
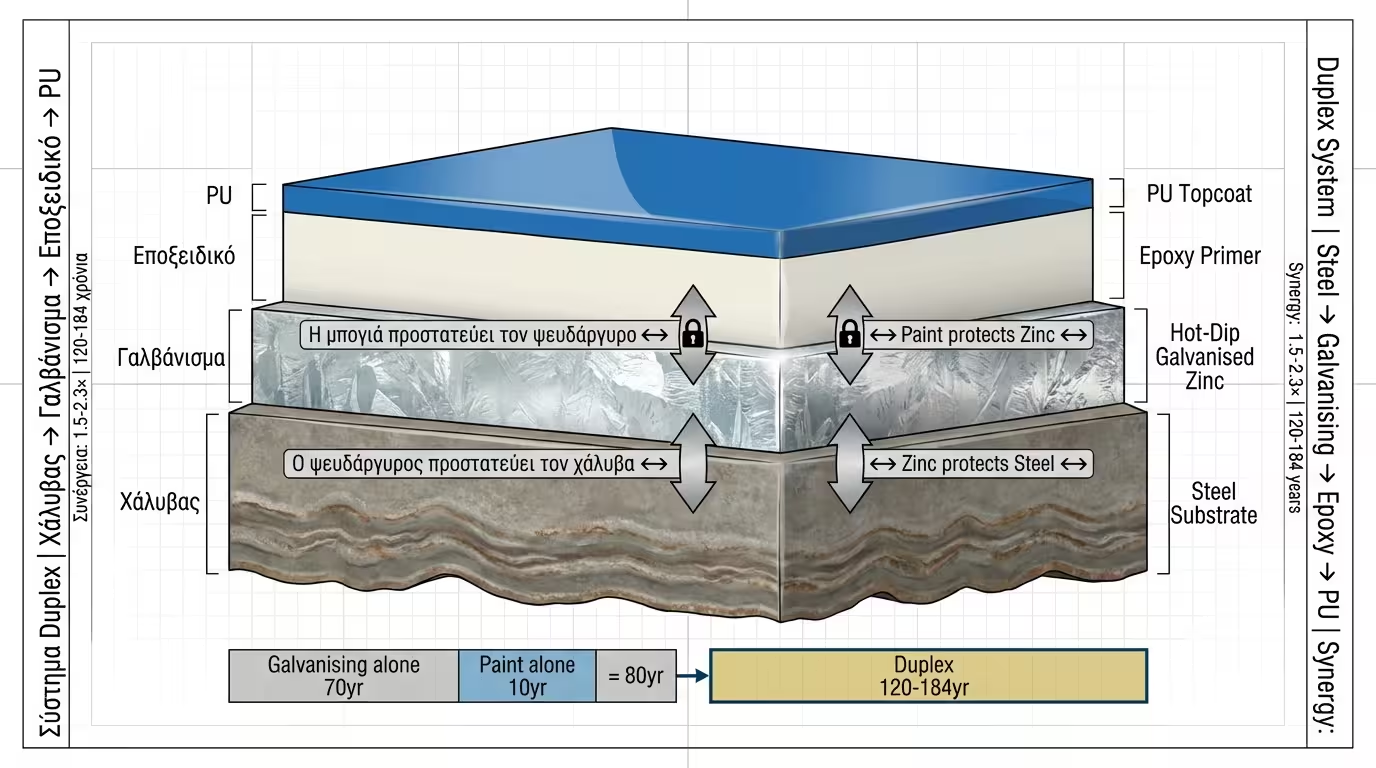
The Duplex System (Paint over Galvanising) is the top-tier protection method worldwide.
The Synergy Formula: Ltotal = 1.5 to 2.3 × (Lgalv + Lpaint).
The paint protects the zinc from erosion, and the zinc prevents oxidation
underneath the paint. This results in durability that can exceed 100 years.
1. St 3 Cleaning (metallic sheen).
2. Converter on joints/deep rust.
3. Primer & 2 coats Topcoat.
1. Sa 2.5 Sandblasting.
2. Zinc-Rich Epoxy primer.
3. Polyurethane Topcoat (High Durability).
Implementation of a Duplex System (Hot-dip galvanising + Powder coating or PU).
Return to category.
Go to categoryReturn to the central guide.
Go to guide