🔋 The Galvanic Circuit
Corrosion requires four elements: an anode, a cathode, an electrolyte (moisture), and a metallic path. The paint acts as an insulator that breaks this circuit, preventing ion movement.
Painting metal is not merely an aesthetic intervention but a critical engineering application designed to counter the thermodynamic tendency of metals to revert to their original ore state via oxidation.
Unlike plaster or wood, metal is an electrical conductor. Rust is not simply "dirt" but an electrochemical phenomenon where microscopic galvanic cells (anode/cathode) form on the metal surface in the presence of moisture and oxygen.
Corrosion requires four elements: an anode, a cathode, an electrolyte (moisture), and a metallic path. The paint acts as an insulator that breaks this circuit, preventing ion movement.
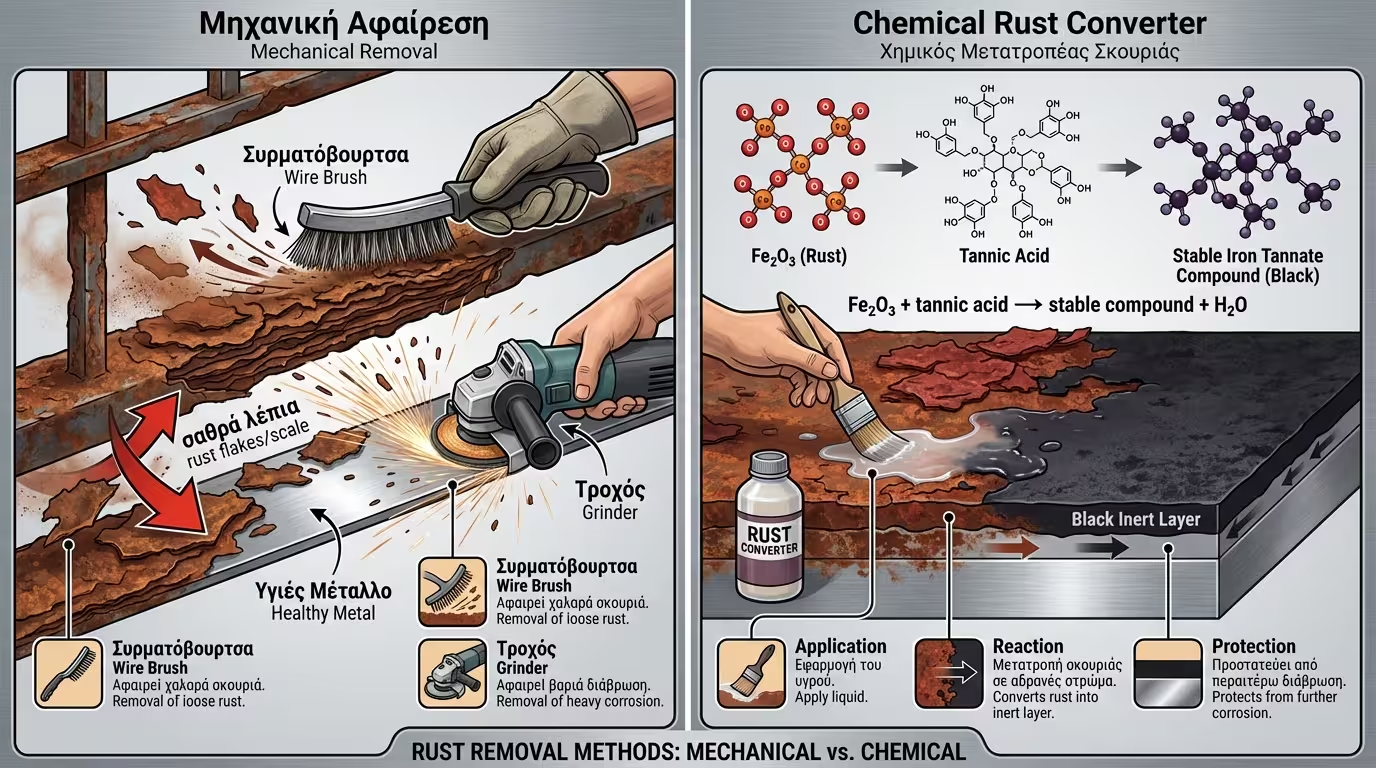
Loose rust must be entirely removed. Professional specifications typically require a Sa 2.5 (Near White Metal) cleanliness level via abrasive blasting, ensuring the primer anchors to a clean crystalline structure.
Metal possesses high surface energy but is often obscured by passivation oils or oxides that reduce it dramatically. Adhesion to metal is primarily chemical and polar, not merely mechanical like on wood.
Steel requires active anti-corrosion protection. Zinc phosphate primers create a passive layer on the metal surface, rendering it chemically inert.
The issue here is surface "instability". A 2-component Wash Primer or specialized adhesion epoxies are required to perform "chemical etching" on the surface, increasing substrate wettability.
Not all paints protect in the same way. The choice of system (primer + topcoat) depends on whether we want to "seal" the metal or protect it electrochemically.
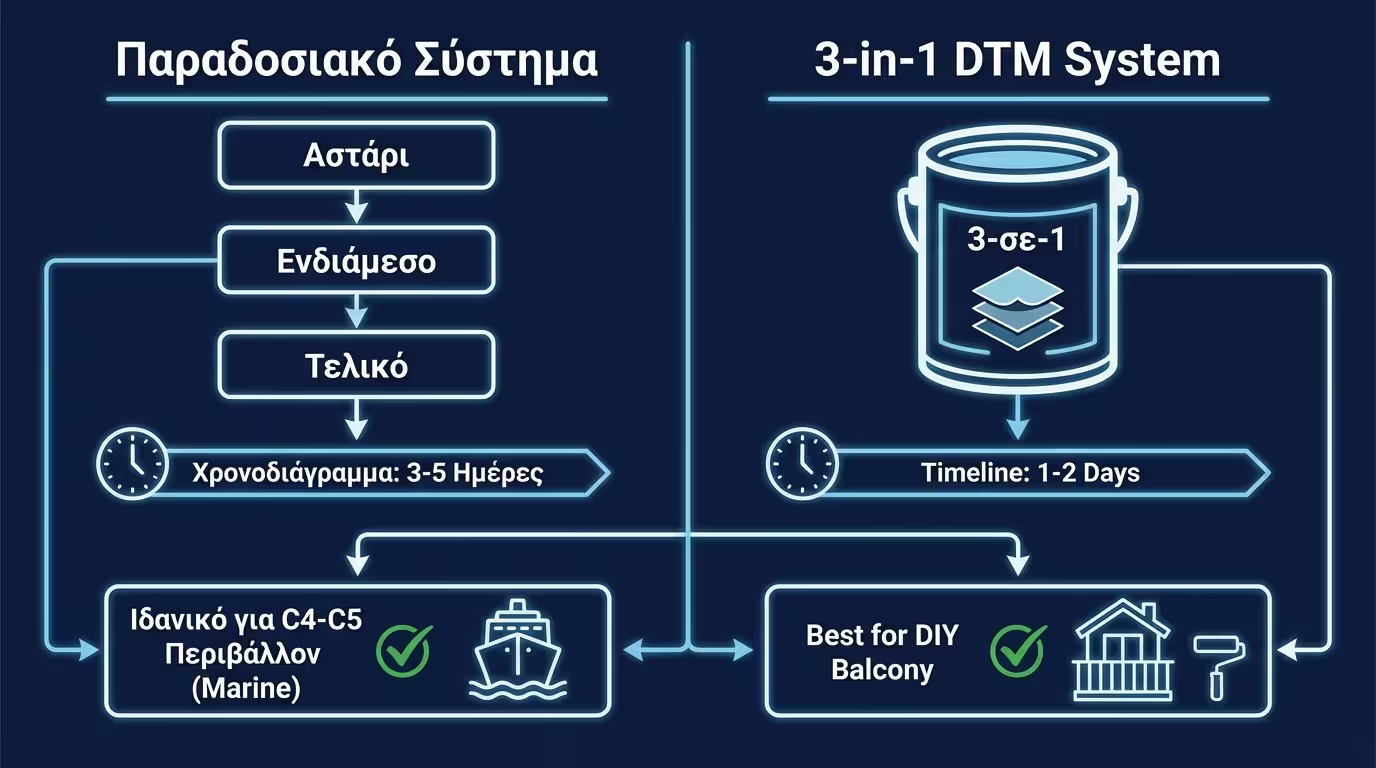
Traditional systems (Primer + Topcoat) rely on film thickness. The "denser" the resin, the harder it is for water and oxygen to penetrate and reach the metal. DFT thickness is key.
In extreme conditions, we use Zinc-Rich primers. Here, the zinc oxidises instead of the iron, providing protection even if the paint is scratched down to the metal.
The global "Bible" of anti-corrosive protection. The ISO 12944 standard now defines 6 categories, with the latest revision introducing CX (Extreme) for offshore structures and tropical environments.

The typical Greek coastal city. Requires systems with DFT > 200μm. A simple single-coat brush application is technically insufficient for a lifespan exceeding 5 years.
How do we verify if the painting "succeeded"? We score a grid into the surface and check for detachment using specialized tape. A Gt0 classification (no detachment) is every professional's goal.
📐 Technical Tip: In metal structures, corners and edges are the first failure points. Always apply a "stripe coat" (an extra brush-applied coat) to edges before full application.
Painting metal is a race against time and chemistry. Investing in a quality system (proper degreasing, appropriate primer, and sufficient DFT thickness) costs less than continuous rust treatment every two years. Remember: paint on metal is not "makeup", it is armour.
Return to category.
Go to categoryReturn to the central guide.
Go to guide